Reaction: Mouse research slows growth of a childhood brain tumour with a very poor prognosis
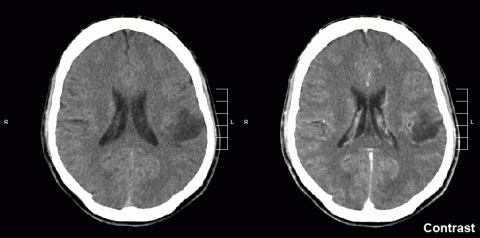
Between 10-15% of childhood brain tumours are diffuse midline gliomas, a type of tumour that occurs within the brainstem, making it almost impossible to remove surgically. Research published in Science Translational Medicine has used a type of molecule (antisense-ASO oligonucleotides) in mouse models of this tumour to slow tumour growth and prolong survival. The authors hope that the research will reach clinical trials and, in that case, the potential new treatment would be combined with others such as radiotherapy or immunotherapy with CAR-T cells.
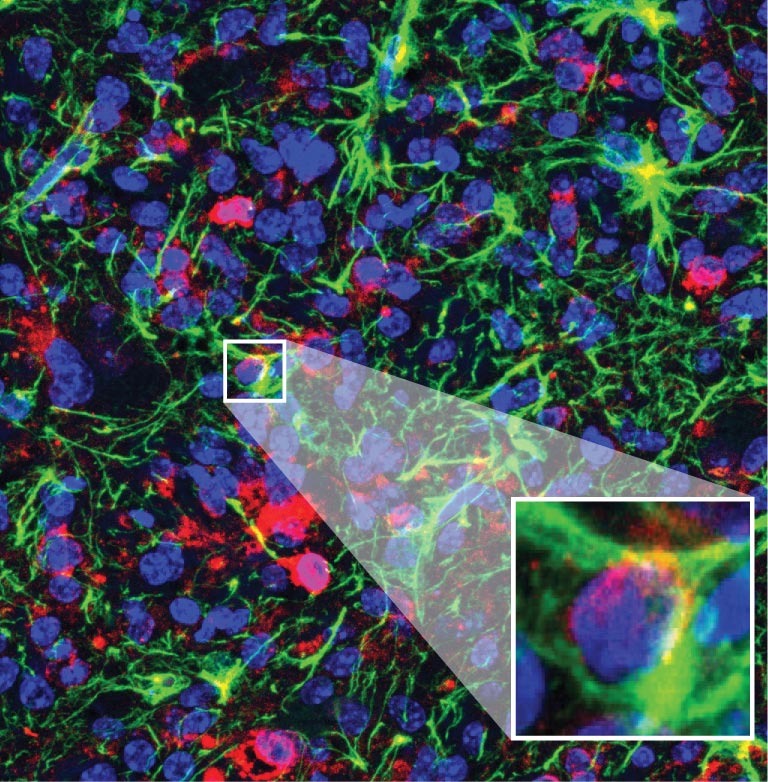
Close-up of diffuse midline glioma tumour cells transforming into healthy cells after treatment with the potential new ASO treatment. Authors: Krainer lab/Cold Spring Harbor Laboratory.
Pilar Sánchez Gómez - tratamiento potencial cáncer cerebral niños EN
Pilar Sánchez Gómez
Senior Researcher at the Carlos III Health Institute, Head of the Neuro-oncology Unit at the Chronic Diseases Research Unit (UFIEC-ISCIII)
This is a study that offers some hope for patients with diffuse midline gliomas and their families. These are tumours with a very poor prognosis that occur in children, adolescents and young adults. Because of their location they are often inoperable and also respond poorly to treatment with radio or chemotherapy.
It has been known for years that most of these tumours express a point mutation in the gene for one type of histone, H3.3, which causes many other downstream genes to be activated and triggers tumour formation. So far, researchers have devised strategies to curb the consequences of this mutation in the H3.3 gene, some of which are even being tested in clinical trials, but so far none have been approved.
In this study, researchers have tried a more direct approach, using specific oligonucleotides to 'break' the mutant gene and inhibit its tumourigenic function, using so-called 'antisense oligos'. This strategy is to some extent similar to that used to generate covid-19 vaccines, but is based on a DNA sequence instead of RNA, which makes it more stable and with modifications that improve its function and its ability to enter tumour cells. In fact, the authors have used the same type of molecules to cure spinal muscular atrophy, demonstrating that they can reach brain tissue. These molecules have already been approved for use in patients in the US and have started to show good results, not only delaying the disease, but also curing some symptoms.
This is a high-quality study conducted at a prestigious centre. In it, the authors conduct an exhaustive search for the most effective antisense oligosense against the mutation present in childhood gliomas and then demonstrate their effectiveness in tumour cells and in mouse models of the disease. For the latter, they make a single injection of the molecules directly into the cranial cavity of the mice, causing the tumours to grow more slowly, but without curing the animals. The models used are relevant, although the authors themselves recognise that the study should be extended to more models that show growth more similar to that of patients.
Also, as the authors point out, the use of these molecules will most likely have to be combined with conventional treatments or more novel therapies, such as immunotherapy. They also comment that they are looking for ways to make this strategy even more effective. However, given the limited therapy options available to patients with this type of glioma and the few side effects that these molecules appear to have, it is not out of the question that they could lead to clinical trials in patients in the medium term.
Declares no conflict of interest in this study.
John E. Wilkinson et al.
- Research article
- Peer reviewed
- Animals