The EU considers the risk of mpox spreading to the general population ‘low’ and does not rule out more imported cases
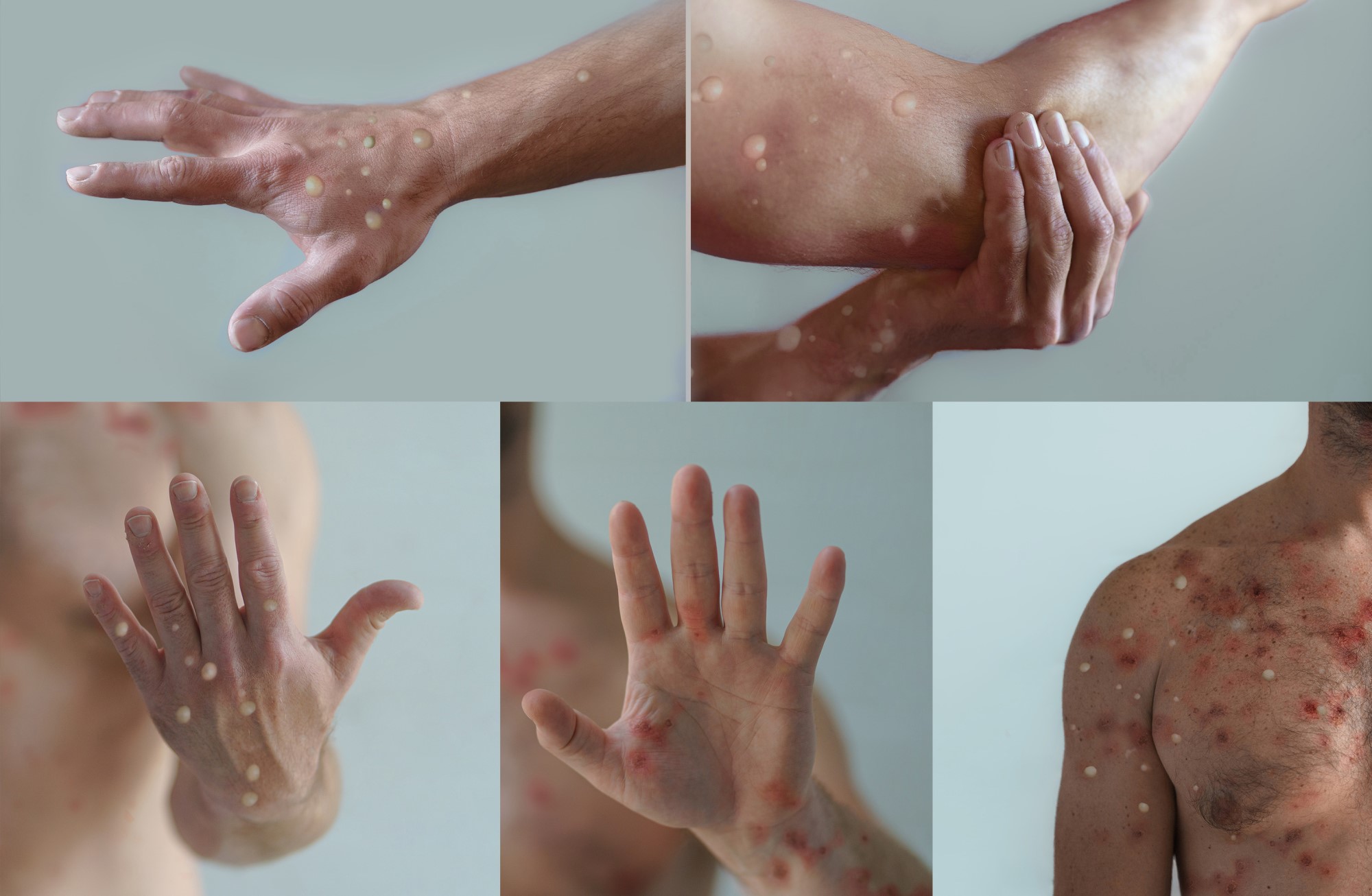
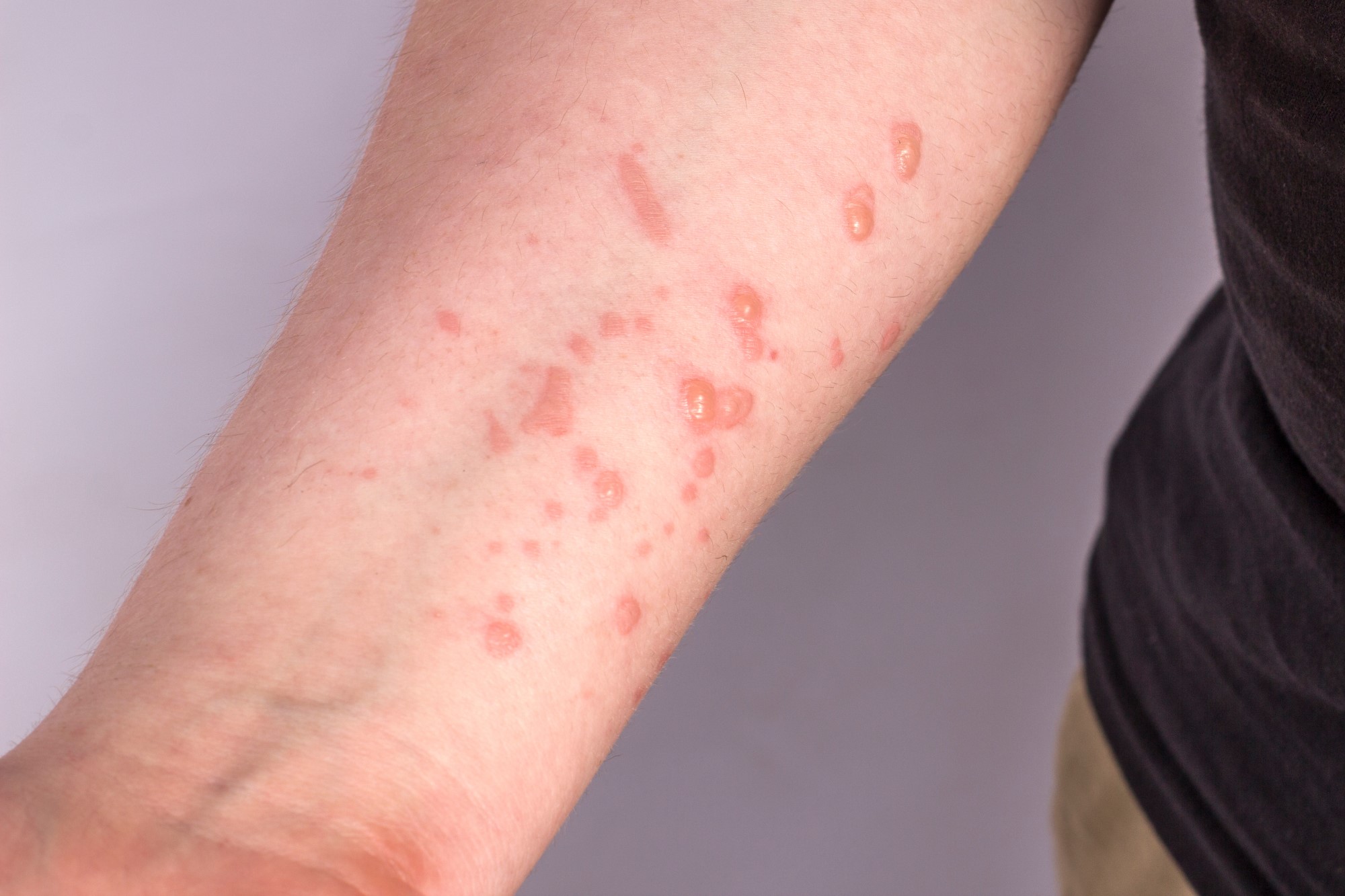
With the increase in cases of mpox, the European Union's Health Security Committee met on Monday. According to the Ministry of Health, the meeting concluded ‘with recommendations very similar to those previously issued by the WHO and the ECDC’. The risk of the likelihood of mpox spreading to the general EU population is currently considered to be ‘low’, although it increases in populations with close contact with diagnosed cases and in particularly vulnerable people. Further imported cases in the EU are not excluded and vaccination of the general population is not recommended. In addition, the HSC does not recommend border control.