The WHO has declared the Ebola outbreak in the Democratic Republic of the Congo and Uganda a public health emergency of international concern
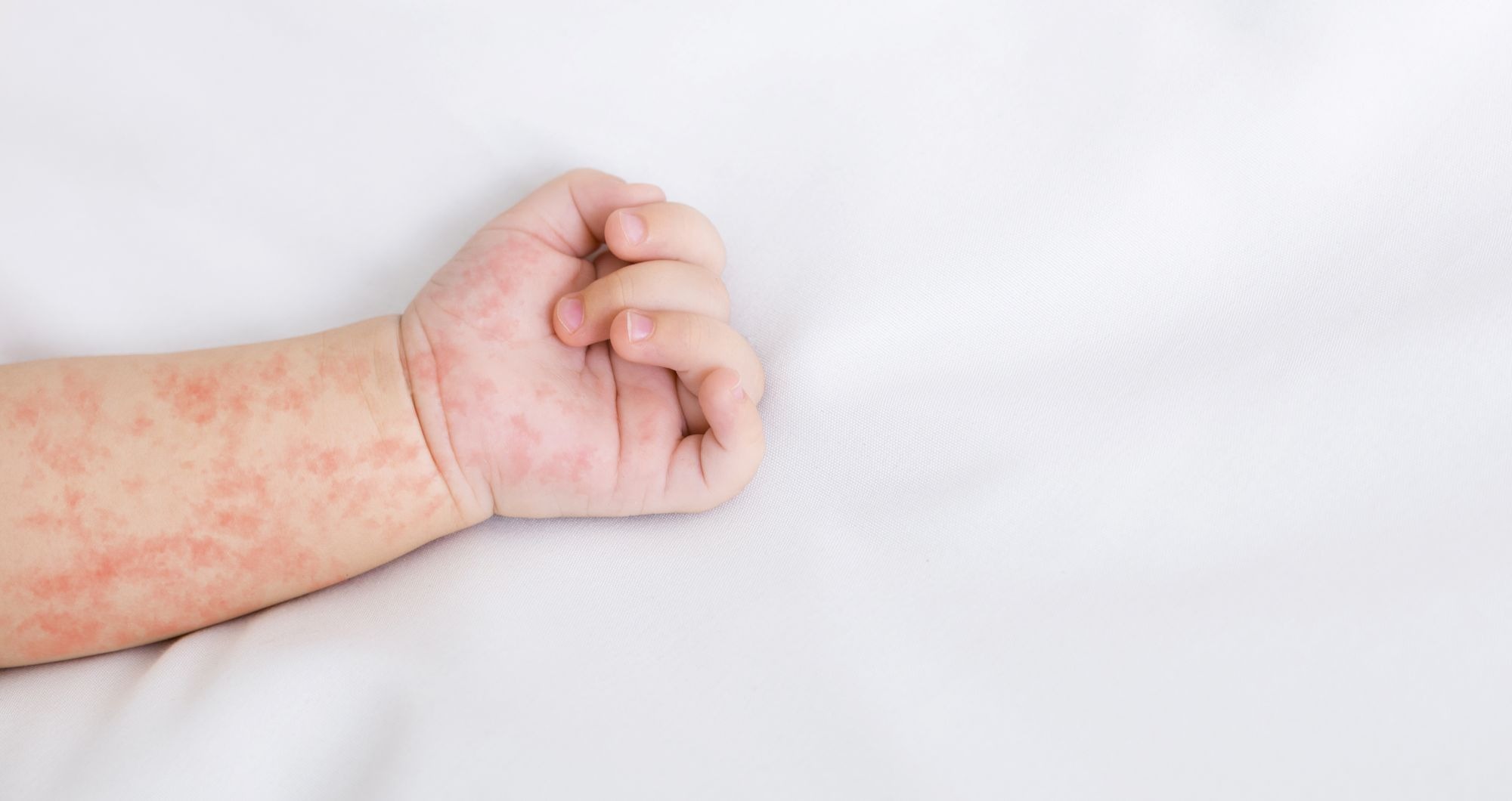
Last Saturday, the Director-General of the World Health Organisation (WHO), Tedros Adhanom Ghebreyesus, declared that the Ebola outbreak caused by the Bundibugyo virus in the Democratic Republic of the Congo and Uganda constitutes a public health emergency of international concern (PHEIC), following consultation with the organisation’s member states. By 15 May, 246 suspected cases and 80 deaths had been reported in three health zones: Rwampara, Mongbwalu and Bunia.